
IT Solutions for Retail and the Consumer Goods Industry
Successful Retail(ing) Through Digital Transformation and Innovation
We Support Your Value Creation Process in Modern Retail
Digitization is an important and pioneering task for retail companies and consumer goods manufacturers: While inflation reduces customers' willingness to buy, the prices of energy and raw materials are rising. In addition, there are tight supply chains, a shortage of skilled workers, regulations relating to working conditions or product safety, and the protection of customer data. Efficient solutions can prevent a digitalization backlog and replace outdated IT systems with modern tools or automation processes - so you are well-prepared for your challenges. Invest in modern IT solutions along your value creation process and gain resources to strengthen your customer loyalty and brand long-term.

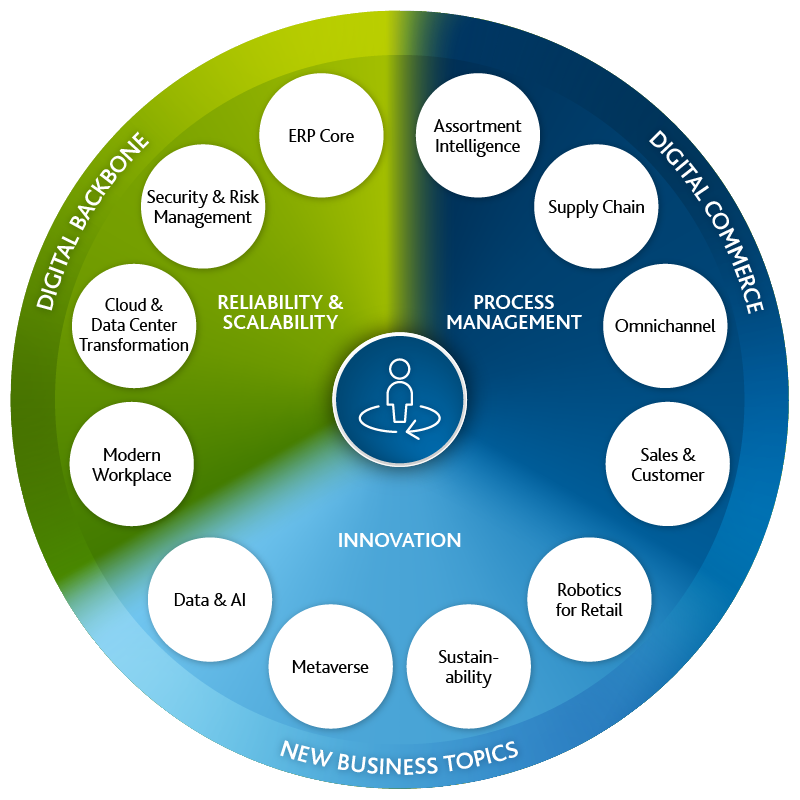
No matter where you currently stand in your digitization strategy: We know the challenges you face and will walk the path of Digital Transformation with you as your consultant, implementation partner and business operations manager. A strong Digital Backbone will ensure a scalable and reliable IT architecture, while industry-specific solutions optimize your processes and enable you to move into innovative business topics.

As with all large projects, there were various, sometimes competing, project goals in this project. But thanks to the professionalism and flexibility of the entire project team, the project success was achieved on time, in quality, and within budget. In this environment, Lekkerland is well-positioned for further growth.
Extract from Our References
Certifications
Rely on us: information security, quality management, IT service management. For our customers, we regularly have our performance measured and extensively certified. You benefit in the long term from our adherence to the highest quality standards and our technological expertise in the form of a trusting and highly professional collaboration.
Worth Knowing about the Digital Transformation in Retail and the Consumer Goods Industry
-
What does Cloud Transformation mean for retail?
Cloud Transformation can bring enormous benefits for the retail and consumer goods industry. By using cloud-based IT solutions, companies can optimize their processes, reduce costs and respond more flexibly to market changes. Through the cloud, data and applications can be centralized, enabling better collaboration within the company. In addition, employees can access the information they need from anywhere, which increases workflow efficiency.
The cloud also offers many benefits for customers. For example, personalized offers can be created by collecting and analyzing data about customers' buying behavior and preferences. Overall, Cloud Transformation can help companies in the retail and consumer goods industry become more competitive and operate more successfully.
-
Why is an Omnichannel Strategy so important?
Today, a successful Omnichannel Strategy is essential for companies in the retail and consumer goods industries. Customers expect a seamless shopping experience across multiple channels, whether online or offline. A well-designed omnichannel strategy enables companies to better understand their customers and provide them with personalized offers. In addition, orders can be processed more efficiently. By processing orders via a Single Source of Truth, customers can be supplied more quickly and receive their products promptly. This leads to higher customer satisfaction, as they can rely on a smooth process.
-
Mastering challenges in logistics sustainably - How is that possible?
One of the biggest challenges in the retail and consumer goods industry is logistics. There are many factors that need to be taken into account here, such as transport costs, warehousing and delivery times. Sustainability in logistics is also becoming increasingly relevant. The aim here is to make transport routes and processes as efficient as possible in order to reduce emissions and conserve resources. Companies can meet these challenges by relying on various IT solutions.
One example is a Warehouse Management System (WMS), which helps to manage inventory. With this system, companies can better control their inventories, which can lead to a reduction in excess inventory and an improvement in delivery times.
Another example is a Transportation Management System (TMS), which helps plan and optimize transportation routes. By using this system, companies can save money while ensuring that products are delivered on time. In addition, real-time data analysis tools can also be used to gain insight into operations and respond quickly to changes.
-
Why is Metaverse the next step Into the future for retailers?
By integrating Augmented Reality (AR) and Virtual Reality (VR), products can be brought to life and customers can test and try them out in a realistic environment. In addition, the Metaverse also offers opportunities for personalized shopping experiences by allowing customers to create individual avatars and thus move around in a digital world.
The topic of sustainability can also be presented and communicated in an appealing way through the Metaverse. By being able to create virtual worlds and implement sustainable concepts in them, companies have the opportunity to showcase their sustainability efforts through the metaverse. For example, they could set up a virtual factory in which visitors can see how resource-saving production processes are carried out.
-
How to maximize Digital Experience and Customer Loyalty?
Customer Loyalty and a unique Customer Experience are not only a key factor for success in retail and for the consumer goods industry, they are also essential for competitiveness. To increase Customer Loyalty, companies must also create a strong brand identity. One way to improve usability and visual quality is to use modern web technologies. State-of-the-art design can also be used to support branding so that it is easily recognizable.
Mobile technologies can also be used to offer users instant access to products and services, as well as live support. This can help your audience solve problems faster, building trust. This also applies to the implementation of personalized recommendation systems. These analyze customers' buying behavior and suggest suitable products. This makes customers feel understood and provides them with an individual shopping experience.
-
How can artificial intelligence support retail?
In retail and the consumer goods industry, it is all about increasing efficiency and saving costs - if used correctly, artificial intelligence can therefore provide a retail company with real added value in the long term. Here are a few examples: AI can continuously optimize product data in e-commerce by detecting and preventing errors in the data. AI-supported demand forecasts can also be used to optimize inventory management and warehouse management. In addition, the customer approach can be automated and personalized with AI and thus achieve higher conversion rates in marketing.
Advantages of AI for your industry at a glance:
- Retail: Optimization of stock levels, personalized shopping experiences and maximum customer loyalty.
- Wholesale: Efficient supply chain management and demand forecasting.
- Manufacturing trading companies: Automation of production processes and quality management.
Your Contact for Retail & Consumer Goods








































